Somatosensory modulation of salivary gene expression and oral feeding in preterm infants: Randomized controlled trial
Despite numerous medical advances in the care of at-risk preterm neonates, oral feeding still represents one of the first and most advanced neurological challenges facing this delicate population. Objective, quantitative, and noninvasive assessment tools, as well as neurotherapeutic strategies, are greatly needed in order to improve feeding and developmental outcomes. Pulsed pneumatic orocutaneous stimulation has been shown to improve nonnutritive sucking (NNS) skills in preterm infants who exhibit delayed or disordered nipple feeding behaviors. Separately, the study of the salivary transcriptome in neonates has helped identify biomarkers directly linked to successful neonatal oral feeding behavior. The combination of noninvasive treatment strategies and transcriptomic analysis represents an integrative approach to oral feeding in which rapid technological advances and personalized transcriptomics can safely and noninvasively be brought to the bedside to inform medical care decisions and improve care and outcomes.
The study aimed to conduct a multicenter randomized control trial (RCT) to combine molecular and behavioral methods in an experimental conceptualization approach to map the effects of pulsed somatosensory stimulation on salivary gene expression in the context of the acquisition of oral feeding habits in high-risk human neonates. The aims of this study represent the first attempt to combine noninvasive treatment strategies and transcriptomic assessments of high-risk extremely preterm infants to (1) improve oral feeding behavior and skills, (2) further our understanding of the gene ontology of biologically diverse pathways related to oral feeding, (3) use gene expression data to personalize neonatal care and individualize treatment strategies and timing interventions, and (4) improve long-term developmental outcomes.
A total of 180 extremely preterm infants from three neonatal intensive care units (NICUs) will be randomized to receive either pulsed or sham (non-pulsing) orocutaneous intervention simultaneous with tube feedings 3 times per day for 4 weeks, beginning at 30 weeks postconceptional age. Infants will also be assessed 3 times per week for NNS performance, and multiple saliva samples will be obtained each week for transcriptomic analysis, until infants have achieved full oral feeding status. At 18 months corrected age, infants will undergo neurodevelopmental follow-up testing, the results of which will be correlated with feeding outcomes in the neo- and post-natal period and with gene expression data and intervention status.
Cortical adapation to orofacial somatosensory stimulation in children and adults
 The integrity of the cerebral cortex can be assessed by measuring its responsiveness to repetitive sensory and motor stimulation. This neurophysiologic feature is known as neural adaptation, and is thought to enhance learning and detection of environmental stimuli. The adaptation of hemodynamic responses to motor and sensory experiences in hand and face are of particular interest—as these are structures most commonly used in human communication—and proper delivery of oxy-hemoglobin to primary motor (M1) and somatosensory (S1) cortices is essential for functional cortical activation.
The integrity of the cerebral cortex can be assessed by measuring its responsiveness to repetitive sensory and motor stimulation. This neurophysiologic feature is known as neural adaptation, and is thought to enhance learning and detection of environmental stimuli. The adaptation of hemodynamic responses to motor and sensory experiences in hand and face are of particular interest—as these are structures most commonly used in human communication—and proper delivery of oxy-hemoglobin to primary motor (M1) and somatosensory (S1) cortices is essential for functional cortical activation.
The goal of the current research project is to examine the hemodynamic differences between hand and face cortical representations during motor and passive somatosensory conditions in children and adults, as measured with functional near-infrared spectroscopy (fNIRS). The recorded data will be used to map developmental changes in patterns of cortical adaptation and hemodynamic responses in an effort to provide a longitudinal picture of normal physiologic connectivity and function, as well as create a computational model of cortical adaptation for future neurodiagnostic and neurotherapeutic applications in disordered populations across the lifespan.
Somatosensory and Motor Function in Individuals with Cerebral Stroke Following Patterned Pneumocutaneous Stimulation

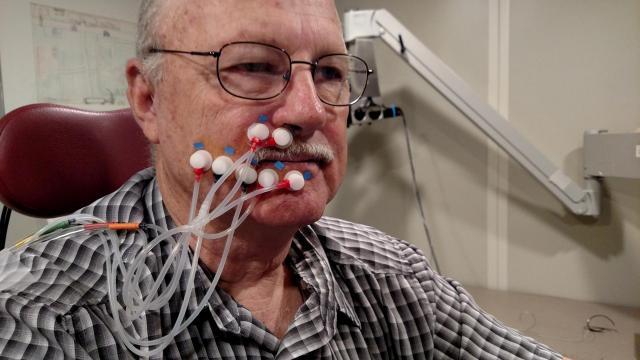
This ongoing study involves approximately 30 adult participants diagnosed with chronic, single hemisphere cortical stroke, and is intended to expand our understanding of the effects of sensory stimulation on sensorimotor impairment in adults who have survived cortical infarct. This is a collaborative project involving researchers and clinicians from UNL and Madonna Rehabilitation Hospital. This study will focus on how neural adaptation induced by sensory training affects sensory and fine motor performance in patients with residual hemiparesis.
Neuroimaging: Velocity Encoding of Saltatory Somatosensory Inputs in Human Brain
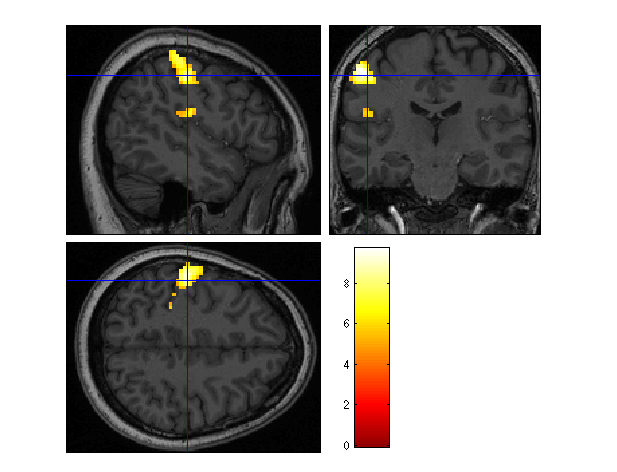
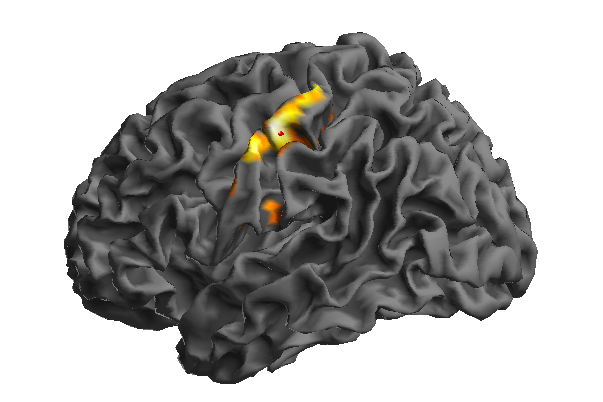
Animal and human models of brain plasticity have shown that the development of functional motor tasks can be facilitated by repetitive sensory stimulation. This development model can be used for the rehabilitation of brain-damaged patients to regain hand movement and speech ability. Published studies of motor tasks have been limited to neurotypical adults using visual and electric stimulation. The aim of the proposed study is to map the relation between saltatory somatosensory (touch pressure) stimulation of the hand and face and activation of primary and secondary somatosensory cortices in neurotypical adults. Saltatory stimulation in this experiment involves the presentation of pneumatic touch pulses which essentially "jump" from one node to another node on the surface of the skin at traverse velocities ranging from very slow to very fast on the surface of the hand. Brain activity will be recorded by placing the participant in the bore of a magnetic resonance imaging (MRI) scanner in order to sample the evoke hemodynamic response to the somatosensory stimulation. Results of this study are expected to lead to the development of innovative neurotherapeutics in adult stroke.
Adaptation of the cSEP Following Pneumatic Stimulation of the Face in Adults*
This study utilized a 4-channel electroencephalography (EEG) recording montage to characterize the adaptation of cortical somatosensory evoked potentials (cSEP) in response to servo-controlled pneumotactile (touch) inputs to the lower face. Stimuli were delivered to 20 healthy adults using a custom built device called the TAC-Cell. This EEG measure of cortical and subcortical adaptation in the neurotypical brain will serve as a referent for future evaluations of disruption associated with acquired (e.g., stroke, TBI) and neurodevelopmental disorders (e.g., ASD).